 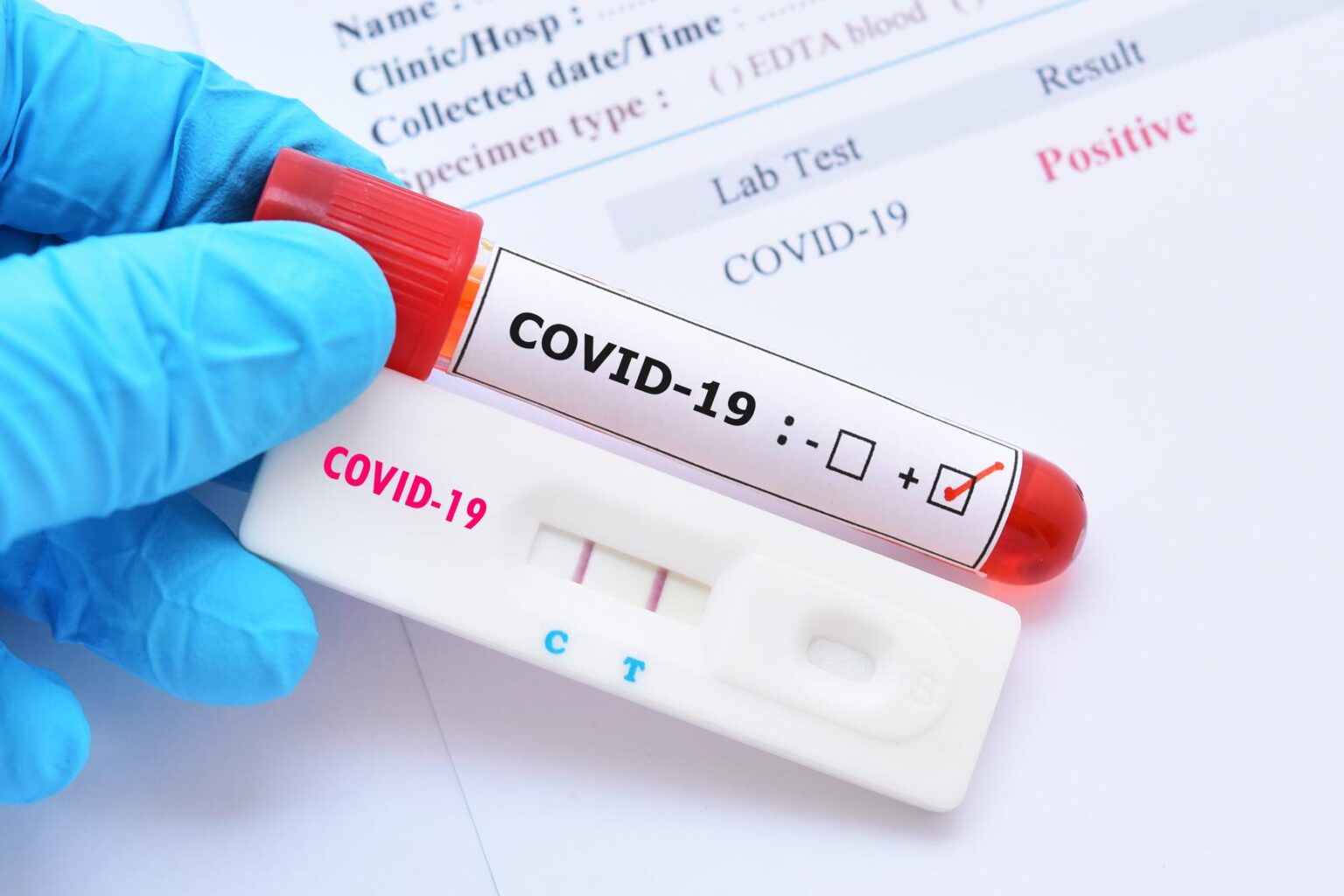 
The BinaxNOW™ COVID-19 Antigen Self Test has not been FDA cleared or approved.*Abbott conducted an analysis of the detection of multiple SARS-COV-2 strains and predicts no impact to the performance of our BinaxNOW COVID-19 Antigen Self Test. 
Or visit the FDA website for the detailed list of extended expiration dates by lot numbers: Please refer to rapidtest.Abbott/BinaxNOW to look up extended dating by lot number. Expiration Date Extension: On December 21, 2022, the FDA granted BinaxNOW™ COVID-19 Self Test 15 month to 22 month shelf-life extension.Serial (or repeat) testing should be performed in individuals with negative results at least twice over three days (with at least 48 hours between tests) for symptomatic individuals and three times over five days (with at least 48 hours between tests) for asymptomatic individuals. Detects Active COVID-19 infection: Includes 2 rapid tests that are indicated for serial testing.For Ages 2 and Above: COVID test suitable for kids, when administered by an adult, and for all people 15 and older to self-administer.Reliable and Easy-to-Use: Requires just a shallow nasal swab that you can do yourself includes easy-to-follow instructions.FDA Authorized 15-Minute Self Test: Convenient COVID-19 at-home test provides fast, reliable results in 15 minutes, results anytime, anywhere no need for a prescription or to send to a lab.Detects multiple COVID-19 variants, including Delta and Omicron*.BinaxNOW™ Self Test uses THE SAME TECHNOLOGY USED BY DOCTORS to test for COVID-19.BinaxNOW™ 2-Test Pack: This double-pack includes 2 at-home tests designed to detect active infection. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
